
Inert gas systems for chemical tankers -Preventing flammable tank atmospheres
In the context of chemical tanker operations and chemical cargoes, an inert gas system may have three distinct uses: preventing
a fire, preventing a chemical reaction or maintaining cargo quality.
Flammable gases normally encountered in chemical tankers cannot bum in an atmosphere which is deficient in oxygen, and an inert gas is understood to be a gas used to produce such an atmosphere by displacing air. SOLAS specifies the standards necessary to do this.
Flammable gases normally encountered in chemical tankers cannot bum in an atmosphere which is deficient in oxygen, and an inert gas is understood to be a gas used to produce such an atmosphere by displacing air. SOLAS specifies the standards necessary to do this.
It may be achieved by using either nitrogen or oil fired flue gas, with a portable or fixed piping
arrangement to supply the inert gas to the cargo tanks and, if applicable, the places surrounding the cargo tanks. Mandatory
safety requirements for tank atmosphere control are given in the IBC Code; for example the system must be able to compensate
for normal transportation losses and maintain an overpressure of at least 0.07 bar gauge at all times.
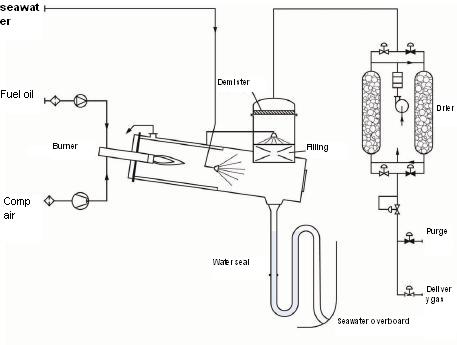
Fig:Inert gas generator
There are several types of inert gas systems that can be used on chemical carriers. The most common are:
There are occasions when inerting is not appropriate for safety reasons, because exclusion of oxygen could create hazardous situations with a number of chemicals when shipped in monomer form. Such chemicals (e.g. acrylic acid, styrene and vinyl acetate) have added inhibitors to prevent polymerisation. during transportation.
In order to be effective, the inhibitors require the presence of oxygen dissolved in the monomer, and that oxygen is obtained from the air in the ullage space. Inhibited monomers must therefore be carried in tanks where the atmosphere has an oxygen level sufficient for the inhibitor to fulfil its purpose.
Requirement of nitrogen used as inert gas on chemical tankers
Most nitrogen used as inert gas on chemical tankers is not used for safety reasons but for cargo quality control. Shippers often have their own special requirements to ensure cargo quality, which can require inert gas of extreme purity, and may specify that nitrogen for initial inerting of cargo systems prior to loading a cargo will be supplied from the loading terminal.
Smaller amounts of pure nitrogen can come from compressed or liquid nitrogen containers stored on board, and refilled from shore when required, but a very high quality can be produced on board by nitrogen generators based on membrane separation, or swing adsorption generators.
When using an oil fired inert gas generator, an oxygen level of less than 5% can generally be obtained, depending on the quality of combustion control and the load on the boiler. The gas must be cooled and scrubbed with water to remove soot and sulphur acids before being supplied to the cargo tanks. But certain cargoes, for instance chemically reactive cargoes, are sensitive to oxygen concentrations as low as 2.0% by volume.
Some cargoes react with carbon dioxide in flue gases. Other cargoes are highly sensitive to moisture, or are liable to discoloration. For these reasons oil fired flue gas systems are rarely used on chemical carriers when carrying chemical cargoes, because demands for strict control of atmosphere standards cannot be met. The following is an indication of potential problems that may occur:
acid catalysed hydrolysis (e.g. with esters, acetates or acrylates);
acid catalysed polymerisation (e.g. with allyl chloride);
formation of carbonates (e.g. with amines);
increased acidity (e.g. contamination of toluene and xylene by carbon dioxide);
reaction with water (e.g. with acetone or ethanol);
de-activation of polymerisation inhibitors (e.g. with vinyl acetate);
Pressure swing Adsorption (PSA) nitrogen generators
Adsorption is a process in which a substance, usually a gas, accumulates on the surface of a solid to form a very thin film. Pressure swing adsorption (PSA) plants work on the principle that the major constituents of air - nitrogen and oxygen - are adsorbed to a different extent when passed over a carbon-molecular sieve material.
The amount of each gas adsorbed depends on the time of exposure. If the system is adjusted correctly, the sieve adsorbs most of the oxygen in the air, allowing the nitrogen to pass through and be collected. The oxygen can then be desorbed (returned to a gas) and exhausted to atmosphere, thereby regenerating the sieve.
To give a continuous nitrogen flow, PSA plants are fitted with two or more interconnected pressurised vessels (called beds) which contain the molecular sieve material. Air is compressed by an oil-free compressor and passed over one set of beds that are adsorbing while the other set of beds is desorbing. During the production cycle, therefore, the plant will vent an oxygen-rich waste, which must be exhausted to a safe area.
In addition to nitrogen and oxygen, the carbon-molecular sieve material also adsorbs a number of other gases, among them carbon dioxide and water vapour.
In normal circumstances the carbon dioxide content in air is very small, so the presence of carbon dioxide has negligible effect on the plant operation and any carbon dioxide adsorbed is ejected with the waste gases during the desorption cycle.
A number of proprietary sieve materials are water sensitive, and the compressed air must be passed through a dryer to remove most of the atmospheric humiditybefore passing over the beds. In marine service, the air inlet to the PSA beds must always be protecfed from spray. -
The gas produced by the PSA process may have an oxygen content varying between 0. 1 % and 2% by volume depending on the flow rate. Typical plants produce gas with a dewpoint lower than -50'C and a carbon dioxide content of less than 2ppm by volume.
Membrane separation nitrogen generators
Membrane units are based on the fact that different gases permeate at different rates through the walls of a thin, hollow membrane. The 'slow' gases are methane, nitrogen and carbon monoxide, the 'medium' gases are argon and oxygen, and the 'fast' gases are water vapour, hydrogen and carbon dioxide.
The fact that the two main components of air, nitrogen and oxygen, have different permeation rates means they can be separated. The fact that water vapour permeates quickly means that the nitrogen produced is also very dry.
The membrane unit is made up from bundles of thin hollow fibres which give a large wall area for separation. The membrane bundles are enclosed in pressure vessel pipes of about 100 to 200 millimetres diameter; several of these bundles may be arranged in parallel.
Clean compressed air is passed into these bundles where the oxygen and water molecules are removed. The membranes are heat-sensitive and it may be necessary to cool the compressed air before it enters the bundles.
The efficiency of the separation depends on the flow rate through the membranes; a control valve is used to regulate the flow and thereby the oxygen content. The flow is adjusted to give nitrogen of the purity required - typically with an oxygen content variable between 0.1% and 2% by volume, with water and carbon dioxide contents below 5ppm. Oxygen enriched air is vented as a waste gas, which must be exhausted to a safe area.
Oil fired inert gas generators
Oil fired inert gas is generally acceptable for use with petroleum products but it has been found that the quality of the inert gas generated by this type of system is not suitable for use with many chemical products, because it can affect the cargo quality.
It is therefore recommended that when inerting or padding is required by the IBC Code for a particular cargo, nitrogen is used to inert or pad that cargo unless the shipper or supplier has stated that oil fired inert gas is acceptable for such purposes.
The basic principle of oil fired plants is that the oxygen content of the air is converted to carbon dioxide by combustion of oil while the nitrogen content remains largely unchanged.
The oil fuel is burnt in a combustion chamber and the combustion (or flue) gas is passed through a water tower (or scrubber) to cool it and remove most of the sulphur dioxide, particulates and impurities. This requires contact between the flue gas and large quantities of sea water.
The gas may then be dried by being passed either through a cooler or an alumina bed dryer (or even both). Chemical tankers are usually fitted with two non-return valves in series as an equivalent to a deck water seal, thereby avoiding the risk of water carry over into the cargo. As a further safeguard against backflow, there is usually an isolating valve or a spool piece at each branch connection.
The inert gas produced by oil fired generators depends on the quality of the fuel oil and the efficiency of the combustion and scrubbing processes. These factors influence, for example, the amount of sulphides in the inert gas produced - which is why the sulphur content of the fuel is limited in the plant specification. Likewise, inefficient combustion can cause soot, which clogs the scrubber and, in particular, the dryer system, thereby producing wet and dirty inert gas.
If the plant is efficiently burning good quality fuel, the inert gas can be expected to have approximately the following composition:
Carbon dioxide 15%
Oxygen 1.0%
Carbon monoxide 0.1%
Oxides of nitrogen 120ppm
Hydrogen 100ppm
Sulphur dioxide and sulphur trioxide 120ppm
Nitrogen Balance
Dewpoint -25'C
Related Info:
Failure prevention and maintenance guideline for inert gas systems onboard
In the event of failure of the inert gas system or supply, action must be taken immediately to prevent air from being drawn into the tanks. All cargo operations must be stopped and the deck isolating valves for the inert gas system closed. Immediate action must be taken to repair the system. & Terminal notified. ....
Cargo tanks inerting safety practice
The term inerting/purging generally refers to the replacement of air in a cargo tank by an inert gas, in chemical tankers most often by nitrogen, in order to prevent the formation of flammable vapours, oxygenation of the product, reduce humidity in the tank and/or protect the quality of the cargo. .....
PV valves operation and maintenance procedure
Pressure/Vacuum valves are designed to provide protection of all cargo tanks against over/under pressure and provide for the flow of small volumes of tank atmosphere resulting from temperature variations in the cargo tank(s) and should operate in advance of the pressure/vacuum breaker, where IG system is in use.
Reference Publications :
Main Info pages!
Home page ||| Chemical hazards ||| Cargo planning & Stowage ||| Cargo loading ||| Cargo documents ||| Safe stability ||| Cargo care ||| Preparation for unloading ||| Inert gas systems |||Gas freeing ||| Nitrogen handling ||| Chemical handling Safe practice |||Handling equipments ||| Cargo & Ballast pumps ||| Cargo tanks |||Tank cleaning |||Special cargoes |||Spills emergencies |||Fire protection

Chemicaltankerguide.com is merely an informational site about various aspects of chemical tankers and safety tips that may be particular value to those working in: Chemical Handling, Chemical Storage, Liquefied Chemical Suppliers, Chemical Shipping, Chemical Transportation, Chemical Terminals, Bulk Chemical Services and Chemical Processing. If you are interested in finding out more about chemical tanker guideline please visit IMO official website. For any comment please Contact us
Copyright © 2011 Chemical Tanker Guide.com All rights reserved.

Fig:Inert gas generator
There are several types of inert gas systems that can be used on chemical carriers. The most common are:
- stored compressed nitrogen;
- stored liquid nitrogen;
- gaseous nitrogen supplied from shore;
- nitrogen generators using pressure swing adsorption (PSA);
- nitrogen generators using membrane separation;
- oil fired inert gas generators.
There are occasions when inerting is not appropriate for safety reasons, because exclusion of oxygen could create hazardous situations with a number of chemicals when shipped in monomer form. Such chemicals (e.g. acrylic acid, styrene and vinyl acetate) have added inhibitors to prevent polymerisation. during transportation.
In order to be effective, the inhibitors require the presence of oxygen dissolved in the monomer, and that oxygen is obtained from the air in the ullage space. Inhibited monomers must therefore be carried in tanks where the atmosphere has an oxygen level sufficient for the inhibitor to fulfil its purpose.
Requirement of nitrogen used as inert gas on chemical tankers
Most nitrogen used as inert gas on chemical tankers is not used for safety reasons but for cargo quality control. Shippers often have their own special requirements to ensure cargo quality, which can require inert gas of extreme purity, and may specify that nitrogen for initial inerting of cargo systems prior to loading a cargo will be supplied from the loading terminal.
Smaller amounts of pure nitrogen can come from compressed or liquid nitrogen containers stored on board, and refilled from shore when required, but a very high quality can be produced on board by nitrogen generators based on membrane separation, or swing adsorption generators.
When using an oil fired inert gas generator, an oxygen level of less than 5% can generally be obtained, depending on the quality of combustion control and the load on the boiler. The gas must be cooled and scrubbed with water to remove soot and sulphur acids before being supplied to the cargo tanks. But certain cargoes, for instance chemically reactive cargoes, are sensitive to oxygen concentrations as low as 2.0% by volume.
Some cargoes react with carbon dioxide in flue gases. Other cargoes are highly sensitive to moisture, or are liable to discoloration. For these reasons oil fired flue gas systems are rarely used on chemical carriers when carrying chemical cargoes, because demands for strict control of atmosphere standards cannot be met. The following is an indication of potential problems that may occur:
acid catalysed hydrolysis (e.g. with esters, acetates or acrylates);
acid catalysed polymerisation (e.g. with allyl chloride);
formation of carbonates (e.g. with amines);
increased acidity (e.g. contamination of toluene and xylene by carbon dioxide);
reaction with water (e.g. with acetone or ethanol);
de-activation of polymerisation inhibitors (e.g. with vinyl acetate);
Pressure swing Adsorption (PSA) nitrogen generators
Adsorption is a process in which a substance, usually a gas, accumulates on the surface of a solid to form a very thin film. Pressure swing adsorption (PSA) plants work on the principle that the major constituents of air - nitrogen and oxygen - are adsorbed to a different extent when passed over a carbon-molecular sieve material.
The amount of each gas adsorbed depends on the time of exposure. If the system is adjusted correctly, the sieve adsorbs most of the oxygen in the air, allowing the nitrogen to pass through and be collected. The oxygen can then be desorbed (returned to a gas) and exhausted to atmosphere, thereby regenerating the sieve.
To give a continuous nitrogen flow, PSA plants are fitted with two or more interconnected pressurised vessels (called beds) which contain the molecular sieve material. Air is compressed by an oil-free compressor and passed over one set of beds that are adsorbing while the other set of beds is desorbing. During the production cycle, therefore, the plant will vent an oxygen-rich waste, which must be exhausted to a safe area.
In addition to nitrogen and oxygen, the carbon-molecular sieve material also adsorbs a number of other gases, among them carbon dioxide and water vapour.
In normal circumstances the carbon dioxide content in air is very small, so the presence of carbon dioxide has negligible effect on the plant operation and any carbon dioxide adsorbed is ejected with the waste gases during the desorption cycle.
A number of proprietary sieve materials are water sensitive, and the compressed air must be passed through a dryer to remove most of the atmospheric humiditybefore passing over the beds. In marine service, the air inlet to the PSA beds must always be protecfed from spray. -
The gas produced by the PSA process may have an oxygen content varying between 0. 1 % and 2% by volume depending on the flow rate. Typical plants produce gas with a dewpoint lower than -50'C and a carbon dioxide content of less than 2ppm by volume.
Membrane separation nitrogen generators
Membrane units are based on the fact that different gases permeate at different rates through the walls of a thin, hollow membrane. The 'slow' gases are methane, nitrogen and carbon monoxide, the 'medium' gases are argon and oxygen, and the 'fast' gases are water vapour, hydrogen and carbon dioxide.
The fact that the two main components of air, nitrogen and oxygen, have different permeation rates means they can be separated. The fact that water vapour permeates quickly means that the nitrogen produced is also very dry.
The membrane unit is made up from bundles of thin hollow fibres which give a large wall area for separation. The membrane bundles are enclosed in pressure vessel pipes of about 100 to 200 millimetres diameter; several of these bundles may be arranged in parallel.
Clean compressed air is passed into these bundles where the oxygen and water molecules are removed. The membranes are heat-sensitive and it may be necessary to cool the compressed air before it enters the bundles.
The efficiency of the separation depends on the flow rate through the membranes; a control valve is used to regulate the flow and thereby the oxygen content. The flow is adjusted to give nitrogen of the purity required - typically with an oxygen content variable between 0.1% and 2% by volume, with water and carbon dioxide contents below 5ppm. Oxygen enriched air is vented as a waste gas, which must be exhausted to a safe area.
Oil fired inert gas generators
Oil fired inert gas is generally acceptable for use with petroleum products but it has been found that the quality of the inert gas generated by this type of system is not suitable for use with many chemical products, because it can affect the cargo quality.
It is therefore recommended that when inerting or padding is required by the IBC Code for a particular cargo, nitrogen is used to inert or pad that cargo unless the shipper or supplier has stated that oil fired inert gas is acceptable for such purposes.
The basic principle of oil fired plants is that the oxygen content of the air is converted to carbon dioxide by combustion of oil while the nitrogen content remains largely unchanged.
The oil fuel is burnt in a combustion chamber and the combustion (or flue) gas is passed through a water tower (or scrubber) to cool it and remove most of the sulphur dioxide, particulates and impurities. This requires contact between the flue gas and large quantities of sea water.
The gas may then be dried by being passed either through a cooler or an alumina bed dryer (or even both). Chemical tankers are usually fitted with two non-return valves in series as an equivalent to a deck water seal, thereby avoiding the risk of water carry over into the cargo. As a further safeguard against backflow, there is usually an isolating valve or a spool piece at each branch connection.
The inert gas produced by oil fired generators depends on the quality of the fuel oil and the efficiency of the combustion and scrubbing processes. These factors influence, for example, the amount of sulphides in the inert gas produced - which is why the sulphur content of the fuel is limited in the plant specification. Likewise, inefficient combustion can cause soot, which clogs the scrubber and, in particular, the dryer system, thereby producing wet and dirty inert gas.
If the plant is efficiently burning good quality fuel, the inert gas can be expected to have approximately the following composition:
Carbon dioxide 15%
Oxygen 1.0%
Carbon monoxide 0.1%
Oxides of nitrogen 120ppm
Hydrogen 100ppm
Sulphur dioxide and sulphur trioxide 120ppm
Nitrogen Balance
Dewpoint -25'C
Related Info:
Failure prevention and maintenance guideline for inert gas systems onboard
In the event of failure of the inert gas system or supply, action must be taken immediately to prevent air from being drawn into the tanks. All cargo operations must be stopped and the deck isolating valves for the inert gas system closed. Immediate action must be taken to repair the system. & Terminal notified. ....
Cargo tanks inerting safety practice
The term inerting/purging generally refers to the replacement of air in a cargo tank by an inert gas, in chemical tankers most often by nitrogen, in order to prevent the formation of flammable vapours, oxygenation of the product, reduce humidity in the tank and/or protect the quality of the cargo. .....
PV valves operation and maintenance procedure
Pressure/Vacuum valves are designed to provide protection of all cargo tanks against over/under pressure and provide for the flow of small volumes of tank atmosphere resulting from temperature variations in the cargo tank(s) and should operate in advance of the pressure/vacuum breaker, where IG system is in use.
Reference Publications :
- IBC/BCH Code
- IMO Inert Gas Systems
- Ships Inert Gas Systems Instruction Manual (Approved by Class)
- Ships Procedure and Arrangements Manual (Approved by Class)
- International Safety Guide for Oil Tankers and Terminals (ISGOTT)
- ICS Chemical Tanker safety Guide
- SOLAS
Main Info pages!
Home page ||| Chemical hazards ||| Cargo planning & Stowage ||| Cargo loading ||| Cargo documents ||| Safe stability ||| Cargo care ||| Preparation for unloading ||| Inert gas systems |||Gas freeing ||| Nitrogen handling ||| Chemical handling Safe practice |||Handling equipments ||| Cargo & Ballast pumps ||| Cargo tanks |||Tank cleaning |||Special cargoes |||Spills emergencies |||Fire protection
Chemicaltankerguide.com is merely an informational site about various aspects of chemical tankers and safety tips that may be particular value to those working in: Chemical Handling, Chemical Storage, Liquefied Chemical Suppliers, Chemical Shipping, Chemical Transportation, Chemical Terminals, Bulk Chemical Services and Chemical Processing. If you are interested in finding out more about chemical tanker guideline please visit IMO official website. For any comment please Contact us
Copyright © 2011 Chemical Tanker Guide.com All rights reserved.
